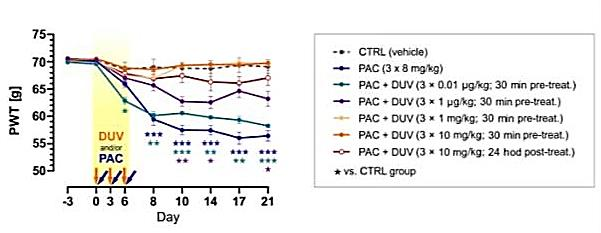
The development of paclitaxel-induced painful peripheral neuropathy (PIPN) is a major side effect of paclitaxel chemotherapy that often leads to limitations in anti-cancer treatment. Our results showed a promising effect of Duvelisib (Copiktra), a novel FDA-approved PI3K isoform-specific inhibitor, in preventing paclitaxel-induced neuropathic pain, reducing pronociceptive signaling in dorsal root ganglion and dorsal horn spinal cord neurons in a model of PIPN. Duvelisib prevented paclitaxel-induced TRPV1 receptor sensitization, reduced PI3K/Akt signaling and reduced number of pathologically increased CD68 cells in spinal ganglia. Specific optogenetic stimulation of inhibitory neurons showed that Duvelisib also blocked the attenuation of inhibitory signaling in the spinal cord in a model of paclitaxel-induced neuropathy. In summary, Duvelisib has demonstrated a promising ability to prevent neuropathic pain in a model of PIPN. The potential application of our findings in human medicine may be enhanced by the fact that Duvelisib is an FDA-approved drug for human use.
Duvelisib treatment (DUV) prevented Paclitaxel (PAC) -induced mechanical hypersensitivity in rats.
Dual PI3Kδ/γ Inhibitor Duvelisib Prevents Development of Neuropathic Pain in Model of Paclitaxel-Induced Peripheral Neuropathy Pavel Adamek, Mario Heles, Anirban Bhattacharyya, Monica Pontearso, Jakub Slepicka, Jiri Palecek J Neurosci. 2022 Jan 10;JN-RM-1324-21, PMID: 35042769 DOI: 10.1523/JNEUROSCI.1324-21.2021
This paper was also selected as Editors pick of the week at the Pain Research Forum. The IASP Pain Research Forum is a production of the International Association for the Study of Pain.